Unveiling the electrical and magnetic properties of lanthanum nickelates
The rare earth nickelates RNiO3 with the high Ni3+ oxidation state have continued to attract enormous interest due to the famous metal-insulator transition and unusual charge and spin-order phenomena. The prediction for multiferroicity and even superconductivity in thin film hetero-structures have triggered new intensive research efforts.
However, our theoretical understanding remains limited. Long-standing questions remain, such as why lanthanum nickelates seem to be the only type of rare earth nickelates that stay metallic and paramagnetic down to lowest temperatures.
Theoretically, as we move along the rare earth nickelates series, the octahedral tilts in their structure become smaller with increasing ionic radius. As a result, the bond angles between the nickel and the oxygen atoms become larger, which alters the electronic bandwidth and the magnetic exchange interactions. With lanthanum having the largest ionic radius of the series, it is expected that the strongest antiferromagnetic properties would be observed in lanthanum nickelates. However, so far they do not show any magnetic order at all.
In order to gain a deeper understanding, an international collaboration from the Max Planck Institute for Chemical Physics of Solids in Dresden, the Julich Centre for Neutron Science at Institut Laue-Langevin (ILL) in Grenoble, the National Synchrotron Radiation Research Center (NSRRC) in Taiwan, and the University of Gottingen and University of Cologne, has worked to look more closely at lanthanum nickelates using neutrons.
While hundreds of publications deal with this class of high oxidation state nickel oxide materials, vast amounts of existing literature are based on powder or polycrystalline samples only. This has in turn restricted how we can look to analyse lanthanum nickelates, and the community’s ability to solve the puzzle.
To uncover the true electric and magnetic properties of lanthanum nickelates, the team set out to create single crystals that were large enough for neutron scattering experiments. This is only possible if the material is grown under high oxygen pressure. Using a 150 bar high-pressure mirror furnace in the Max Planck Institute’s Physics of Correlated Matter Department, the team was able to synthesize single crystals that were large enough for neutron scattering experiments.
Using the ThALES, IN8 and IN12 spectrometers at the ILL they were able to observe magnetic excitations in this single crystal. This suite of instruments was chosen for its unmatched high neutron flux and the possibility of performing polarization analysis.
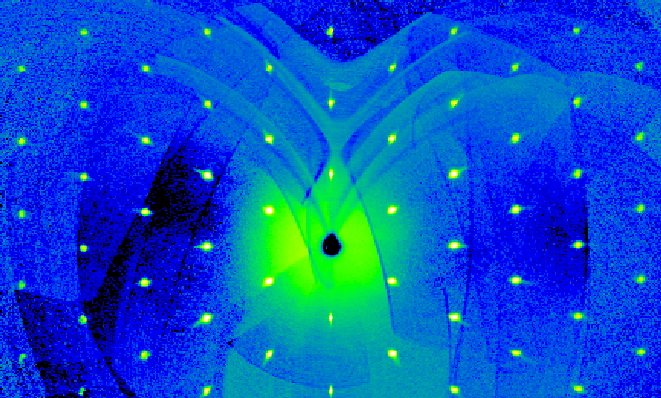 |
Fig. 1 Intensities within the HK0 plane of reciprocal space obtained in a single crystal X-ray diffraction experiment on LaNiO3 |
Some of the results are shown in Figures 1 (png - 147 Ki) and 2 (png - 25 Ki). In addition, the ordering temperature for lanthanum nickelate was found to be relatively high, at 157 Kelvin. The electrical resistivity and Hall effect measurements on the single crystals revealed that lanthanum nickelate is intrinsically not a bad metal, as recently thought for the rare earth nickelate compounds in their paramagnetic phase. On the contrary, the team found that lanthanum nickelate actually has a quite high conductivity. The material appeared to be a highly metallic and antiferromagnetic transition metal oxide, a rather rare combination in transition metal oxides. These characteristics also occur very close to an insulating state, probably close to a quantum critical point, where strong local electronic correlations at the nickelates are likely to interfere in an intricate manner with Fermi surface effects.
As a consequence of these observations, the well-known rare earth nickelate phase diagram was subject to changes, as outlined in Figure 3 (png - 35 Ki). This new result calls for new theories about how to explain the properties and the electronic structure of these high oxidation state materials.
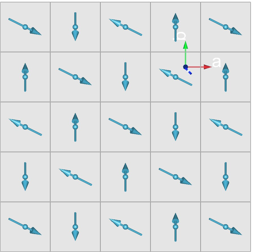 |  |
Fig. 2 A spiral spin arrangement that is consistent with the neutron scattering experiments on | Fig. 3 New tentative phase diagram of the RNiO3 system. |
Re.: Nature communications, volume 9, Article number: 43 (2018). DOI:10.1038/s41467-017-02524-x
Contact: Andrea Piovano, Research Scientist, Spectroscopy Group
