General ILL Seminar, Organised by College 5B - Dr. Sabrina GRENDA
25/10/2024 - 09h00 to 10h00Room 7/8, in ILL 1 building
"Open-shell borazine: Synthesis and magneto-structural relationships"
By Dr. Sabrina GRENDA
Laboratoire des Multimatériaux et Interfaces (LMI, UMR 5615) - Université Claude Bernard Lyon 1, Villeurbanne, 69100
Borazine (B3N3H6) is a heterocyclic compound made of alternating boron and nitrogen atoms, known as the "inorganic benzene". However, despite similarities with benzene, because the C=C and B=N bonds are isostructural and isoelectronic, the borazine ring has very different electronic properties due to significant differences in electronegativity between the boron and nitrogen atoms.
We synthesized the first example of borazine functionalized by nitroxide radicals: the N,N',N''-tris(4‑Bromophenyl))-B,B',B''-tris((2,6-dimethyl-4-(N-tert-butyl-N-oxyamino)phenyl)borazine (simplified as Borazine-B-Tris-Nitroxide), in order to study the exchange coupling of nitroxide radicals via the borazine ring and to compare it with other π-conjugated systems.
In the seminar, we will discuss about the synthetic strategies followed, which enabled us to obtain this first open-shell borazine with high stability. We will see that the molecule exhibits crystalline polymorphism, directly impacting the magnetic properties of these systems. Two phases, labeled a and b, have been isolated and their magneto-structural characterization will be presented. We have completed this work with charge and spin density mapping studies using high-resolution X-ray diffraction and polarized neutron diffraction to understand how unpaired electrons are delocalized under the effect of the borazine core.
Taken together, these studies help to rationalize the relationships between structure, magnetic properties and exchange interactions involving the borazine core for the first time.
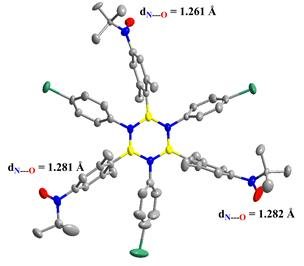
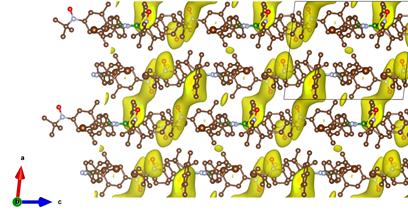
Figure 1 : Cystal structure of Borazine-B-Tris-Nitroxide
Figure 2 : Spin density mapping between the 2D layers using maximum entropy, highlighting the presence of exchange interaction between the nitroxide functions with the shortest intermolecular distances. The iso-contours are set to 0.181 mB
Iurii KIBALIN (College 5 B secretary)
External visitors may ask for a site access to Brigitte Dubouloz (dubouloz@ill.fr)
