Filming the self-assembly of macromolecules in slow motion!
05 Sep 2023In an article published in ACS Macro Letters, a team of physicochemists describes a novel approach which uses neutron and light scattering to monitor in situ the self-assembly of amphiphilic block copolymers in near-equilibrium conditions. These results offer a wealth of possibilities for observing and better understanding the organisation of complex biological and synthetic macromolecules in aqueous solutions.
Though continuous down-scaling in microelectronics has enabled the industry to achieve phenomenal success, the trend is now approaching fundamental limits. Promising alternative technologies for next generation devices exploit not only the charge of the electron – as used in conventional microelectronics – but also the electron spin (spintronics) or spin waves (magnonics) in order to transmit, store and process information with improved energy efficiency, processing speed and memory storage density. Neutrons are a uniquely powerful probe for the advancement of these technologies due to the intrinsic spin of neutrons that provides them with a magnetic moment and thus the capability to reveal not only the atomic-level structure of materials but also the microscopic magnetic structure and dynamics. The Institut Laue-Langevin (ILL) is at the forefront of research in this area, contributing to and benefiting from the world-class magnetism expertise in Grenoble that can be traced back to Louis Néel, whose contributions to magnetism were acknowledged by a Nobel Prize.
For several decades, yttrium iron garnet (YIG) has been the prototypical material used to study and advance research in a variety of spintronic and magnonic domains. This essential material was discovered and its magnetic structure elucidated by Bertaut, Forrat and Néel, working in Grenoble in the 1950s. Neutron diffraction was subsequently used to verify the proposed models and provide a detailed description of YIG’s complex ferrimagnetic structure. “In common ferromagnetic materials, such as iron, the magnetic moments are aligned in the same direction creating a north and south pole,” explains Timothy Ziman, CNRS research director and member of the ILL Theory Group. “YIG, however, is composed of charged iron atoms caged within two structurally different types of interlocking crystal subunits. Overall net magnetisation is created by the 3:2 ratio of these subunits, with the iron ions collectively aligned but in opposite directions.”
Theory had long predicted that magnons propagating in a structurally complex ferrimagnet like YIG split into two branches with opposite polarisations and different energies. This magnon polarisation was explicitly demonstrated in YIG for the first time by an experiment successfully carried out on the polarised neutron inelastic scattering instrument – IN20 – at the ILL, with the results published in Physical Review Letters in 2020. [1] “IN20 is the uncontested best instrument worldwide to study the polarisation of magnons in the thermal energy range,” explains Mechthild Enderle, ILL scientist and first responsible for IN20. “The most original aspect of the experiment,” continues Ziman, “is that the chirality was visible not only near absolute zero temperature, as was expected, but even at room temperature, which could be hugely significant for potential future applications such as spintronics.”
The unique properties of YIG have ensured that it is a well-known and widely used material. In comparison, the other members of the rare-earth iron garnet series have currently very limited use. “It’s an incredibly flexible family of compounds, all chemically very similar and with essentially the same fairly open structure, where yttrium can be replaced by other rare-earths such as terbium, gadolinium, thulium or samarium,” explains Ziman. YIG, though complicated, is the simplest member of the rare-earth iron garnet series due to the fact that the yttrium ion has no magnetic moment, as opposed to the other rare-earth ions which each bear their own magnetic moments. Though the substitution of non-magnetic yttrium ions with magnetic rare-earth ions introduces an additional level of complexity, it is also responsible for interesting properties with strong potential applications. There has thus been a significant renewal of interest in the study of the spin structures and spin wave dynamics of the entire rare-earth iron garnet series.
Terbium iron garnet is one such example for which exploratory experimental data has been acquired at the ILL using IN20. “In terbium iron garnet, the terbium ions are pointing in one direction and the iron ions in the other direction but the overall net moment is dominated by terbium due to their much greater magnetisation, meaning that you’re starting with a ferrimagnet like YIG,” explains Ziman. “As the temperature increases though, terbium starts fluctuating up and down and its average moment gets smaller. At a certain point, referred to as the compensation temperature, the magnetisation moment of the terbium ions and iron ions are exactly balanced and essentially you have, at this one temperature, an antiferromagnet.”
Antiferromagnetism – where the magnetic moments in a material are of equal magnitude but aligned in opposite directions – was first proposed by Néel and later proved using neutrons. Today, antiferromagnets attract significant interest due to their promising applications in the field of spintronics-based magnetic memory. “Uniformly magnetised regions in magnetic materials are called domains and domain walls are the finite regions separating domains and over which the magnetisation rotates from one domain orientation to another,” explains Ziman. “A major challenge in magnetic memory is the speed at which domain walls can be moved and ultrafast domain wall motion has been observed in ferrimagnetic materials near a second compensation temperature at which the net angular momenta of the sublattices cancel.” The spin dynamics at and around the compensation temperatures is thus an active area of research.
It is expected that future polarised neutron inelastic scattering experiments on IN20 at the ILL will cover the entire series of rare-earth iron garnets in order to attain a full quantitative understanding of their spin-wave dynamics. In parallel, theoretical and experimental neutron diffraction studies continue to advance our understanding of the structures of these compounds, first elucidated in Grenoble in the 1950s. A recently published study has investigated the complicated umbrella-like spin structure of rare-earth iron garnets and how it changes with temperature. [2] “At low temperature, terbium iron garnet has three terbium ions which each point in different directions and together form a three-ribbed open umbrella structure. The ribs of the umbrella close, however, as the temperature is raised,” explains Ziman. The detailed information provided by such studies contributes to the interpretation of experimental observations, in addition to the guidance of future experimental work.
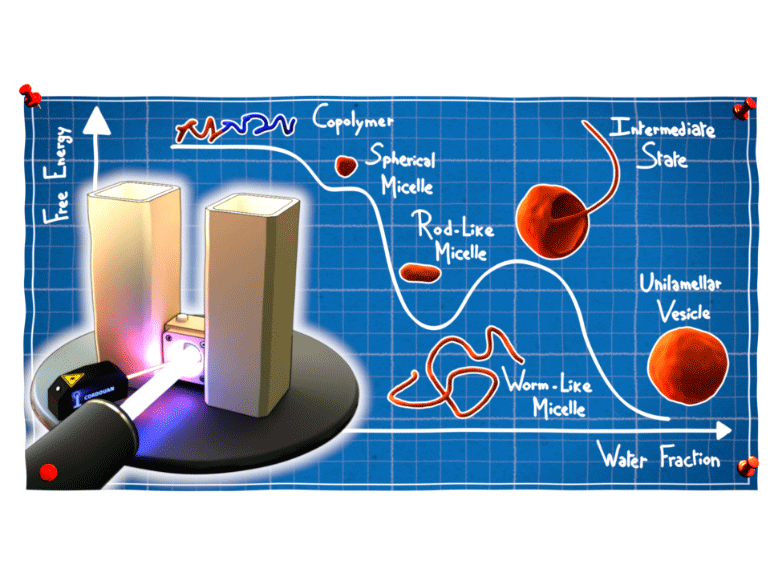
Micelles, vesicles, polymersomes, lipid membranes... these are just some of the self-assembled structures formed by amphiphilic macromolecules. These molecules contain both a hydrophilic (“water-loving”) part and a hydrophobic (“water-fearing”) part and are widely used in everyday life products from paints and pharmaceutical products to food and cosmetics. They make it possible to disperse an oily phase into an aqueous phase and to stabilise nano-objects – and structures with all kinds of geometries – in different media. Their spontaneous self-assembly is driven by weak interactions between the hydrophobic parts, which cluster together to minimise their contact area with water, while the hydrophilic parts repel each other.
The self-assembly mechanisms of amphiphilic macromolecules have been widely studied and are, in theory, predictable, provided they are controlled by thermodynamics. In practice, however, the mobility and reorganisation dynamics of large molecules are very slow. Often, the structures obtained are not the most thermodynamically stable structures, but those which form the fastest.
These non-equilibrium structures depend very much on the experimental conditions in which they are formed, prompting the question:
"how can we slow down this process sufficiently to be able to observe which structures really are the most stable, and monitor the different stages of their formation?"
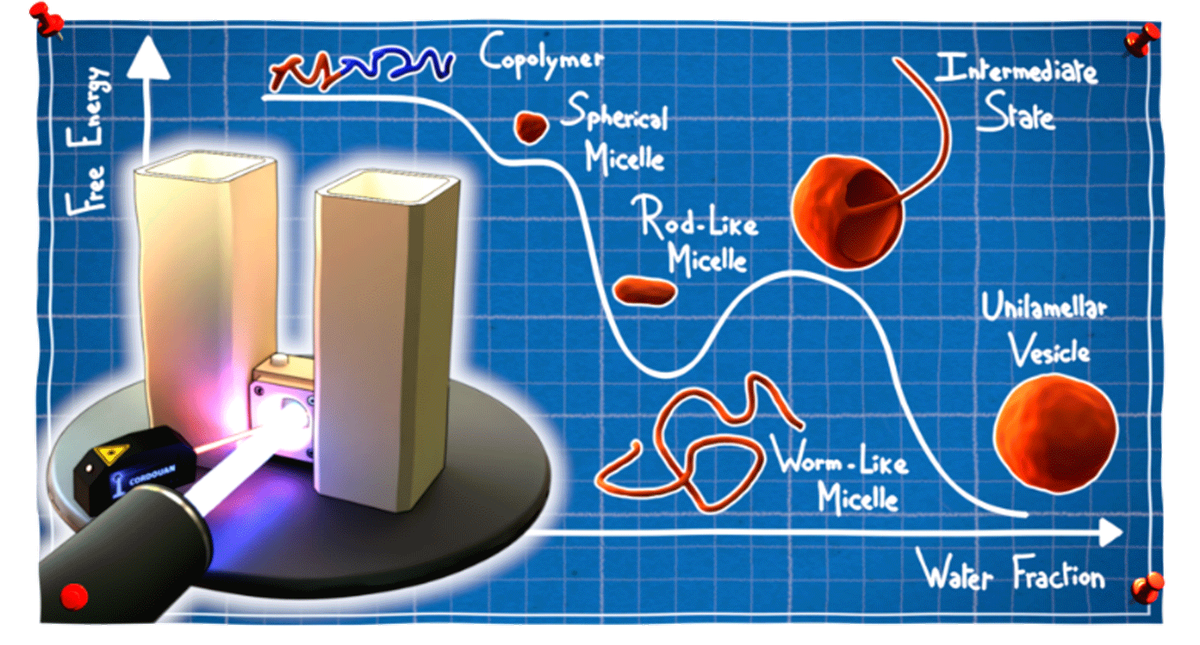
To find the answer to this question, physicochemists expert in soft matter from 4 CNRS laboratories* teamed up with scientists from the Institut Laue Langevin (ILL) to develop an innovative experimental protocol using a dialysis set-up developed at the ILL, which allows water to diffuse slowly in a mixture containing a miscible solvent.
The very gradual increase in the proportion of water slowly activates the attraction between the hydrophobic parts, which in turn triggers the self-assembly of macromolecules in solution. What makes this set-up so unique is that it allows the self-assembly mechanisms to be observed in situ using a combination of dynamic light scattering (DLS) and small-angle neutron scattering (SANS), both of which are suitable for the investigation of nanostructures ranging from 1 to 100 nm. Scientists were therefore able to monitor the self-assembly of chains of amphiphilic diblock copolymers made of poly(ethylene glycol) and poly(dimethylsiloxane) (PEG-PDMS) throughout the process, and establish their phase diagram under quasi-thermodynamic equilibrium. The study was rounded off by cryo-electron microscopy and surface tension analysis of the structures formed.
Presented in ACS Macro Letters, this method can be applied to all types of synthetic and biological macromolecules and colloids. Indeed, the dialysis technique implemented on the large dynamic range SANS diffractometer D22 at the Institut Laue-Langevin enables the slow diffusion not only of solvents but also of any other molecules capable of triggering self-assembly, such as acids or bases, ions, hydrogen bond donors/acceptors and biological ligands. For those in the physical chemistry community seeking to understand the self-assembly mechanisms of these systems, this new technique opens up exciting new prospects.
ILL Instrument: D22, the Large dynamic range small-angle diffractometer
Reference: : https://doi.org/10.1021/acsmacrolett.3c00286
Green Open Access: https://hal.science/hal-03841722
ILL Contact: Lionel Porcar
Contact: Christophe Schatz
* Laboratoire de chimie des polymères organiques (LCPO, CNRS/Bordeaux INP/University of Bordeaux), Centre de recherche Paul Pascal (CNRS/University of Bordeaux), Institut Charles Sadron (CNRS/INSA Strasbourg/University of Strasbourg), Laboratoire Léon Brillouin (LLB, CNRS/CEA)
