SuperADAM
CRG - Advanced Reflectometer for the Analysis of Materials
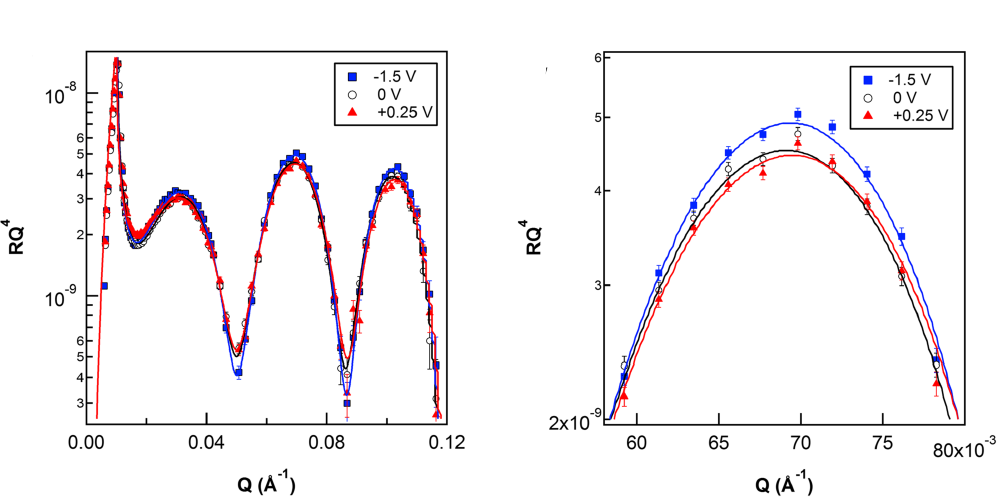
(left) RQ4 vs. Q for 20% w/w [P6,6,6,14][BMB] in acetone-d6 on a gold electrode as a function of the momentum transfer vectorQat different applied potentials. The symbols show experimental data, whilst the solid lines represent one-slab model fits to the data for each potential. The error bars represent the statistical error (standard deviation) in the experimental reflectivity data. (right) The same data presented for a limited Q-range within which the second reflectivity maxima lies.

(left) Normalised reflectivity R near the first maxima as a function of applied potential with respect to the reflectivity measured 0 V. The reflectivities are averages of measurements taken at 5 min intervals over 30 min. The error bars are the relative errors in the intensity calculated by a standard error propagation. The inset shows the same reflectivity data, but as a function of time, indicating the potential bias direction. The solid black lines represent the line of best fits for the data presented. (right) Model SLD profiles obtained from one layer fits to the reflectivity curves for 20% w/w [P6,6,6,14][BMB] in acetone-d6 on a gold electrode. The inset of (right) shows the interfacial region of the SLD profiles on an expanded scale.
Electro-responsivity of ionic liquid boundary layers in a polar solvent revealed by neutron reflectance
Using neutron reflectivity, the electro-responsive structuring of the non-halogenated ionic liquid (IL) trihexyl(tetradecyl)phosphonium-bis(mandelato)borate, [P6,6,6,14][BMB], has been studied at a gold electrode surface in a polar solvent. For a 20% w/w IL mixture, contrast matched to the gold surface, distinct Kiessig fringes were observed for all potentials studied, indicative of a boundary layer of different composition to that of the bulk IL-solvent mixture.With applied potential, the amplitudes of the fringes from the gold-boundary layer interface varied systematically. These changes are attributable to the differing ratios of cations and anions in the boundary layer, leading to a greater or diminished contrast with the gold electrode, depending on the individual ion scattering length densities. Such electro-responsive changes were also evident in the reflectivities measured for the pure IL and a less concentrated (5% w/w) IL-solvent mixture at the same applied potentials, but gave rise to less pronounced changes. These measurements, therefore, demonstrate the enhanced sensitivity achieved by contrast matching the bulk solution and that the structure of the IL boundary layers formed in mixtures is strongly influenced by the bulk concentration. Together these results represent an important step in characterising IL boundary layers in IL-solvent mixtures and provide clear evidence of electroresponsive structuring of IL ions in their solutions with applied potential.
THE JOURNAL OF CHEMICAL PHYSICS 148, 193806 (2018) / doi https://doi.org/10.1063/1.5001551
