D23
CRG - Thermal neutron two-axis diffractometer for single-crystals
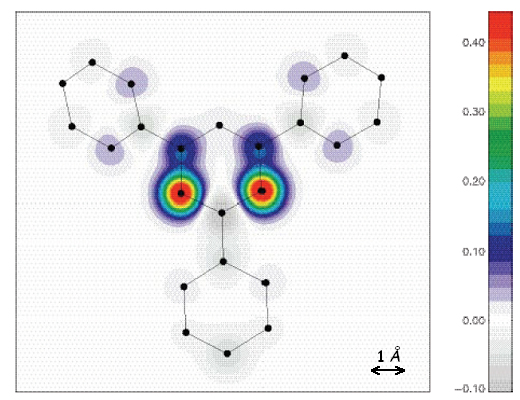
Magnetization distribution and magnetic structure in the free-radical TPV
The organic compound 2,4,6-Tri-PhenylVerdazyl (TPV) is a stable free radical of fomula C20N4H17, built of a verdazyl ring (carrying a spin 1/2) surrounded by three phenyl groups.This compound is described by linear S = 1/2 antiferromagnetic (AF) Heisenberg chains. Below TN = 1.78 K, due to small interchain interactions, a three-dimensional AF ordering develops, described by a k = 0 propagation vector, while a weak ferromagnetic component (of the order of 10-3 μB per molecule) also appears.
The magnetisation distribution inside the molecule has been determined using polarised neutrons. The magnetisation is delocalized on the phenyl ring, with a sign alternation due to the spin polarisation effect, while the four nitrogen atoms are not equivalent: those connected to the phenyl rings carry 0.15(1) μB and the two others carry 0.54(1) μB. The magnetic structure has then been determined, using unpolarised neutrons. The AF ordering is described by a collinear structure with moments of 0.39 μB pointing along the a-axis, direction compatible with a weak ferromagnetism along the c-axis, in terms of group theory. This small AF moment (1 μB is expected for a spin 1/2) can be interpreted interms of spin reduction in S = 1/2 Heisenberg AF chains.
More details in:
Tomiyoshi S., Ressouche E., Schweizer J., Tagami Y., Azuma N. - Magnetic structure of the free-radical 2,4,6-triphenylverdazyl (TPV) Physica B 276-278, 748-749 (2000)