D20
High-intensity two-axis diffractometer with variable resolution
 (jpg - 111 Ki)
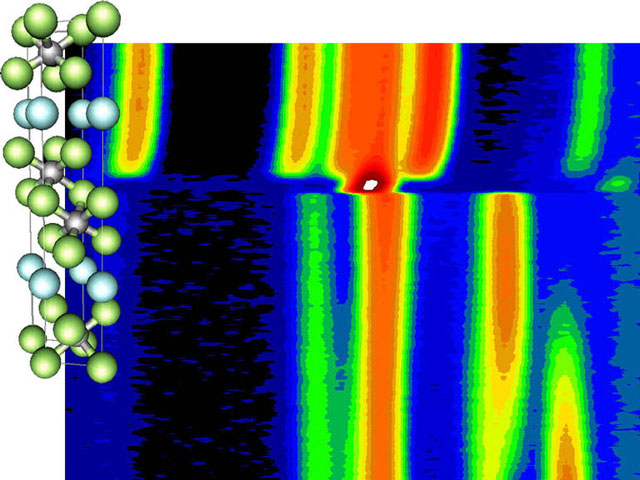
(jpg - 111 Ki)Structure of Ti3SiC2 and time-resolved neutron diffraction patterns recorded on D20Intensity is color-coded, from dark blue for low intensity over green and yellow and finally red and white for high intensity.
Kinetics: Self-propagating high-temperature synthesis (SHS)
In situ neutron diffraction of the SHS of the high-temperature ceramical material Ti3SiC2 (see movie) was performed. Powder diffraction patterns were taken every 0.9 seconds while the precursors were heated from 850°C to 1050°C with 100°C per minute. They (see figure) indicates an α and β phase transition in titanium at 870°C, pre-ignition reactions, the formation of a single intermediate phase in only 0.5 seconds after the ignition, and after a delay of 2 seconds (during excess heating of 2000°C) the rapid nucleation and growth of Ti3SiC2.
The whole reaction is over in only 5 seconds after the ignition. The pre-ignition reactions are exothermic and provide the extra heat for a sustained SHS to occur. No amorphous contribution to the diffraction patterns was detected and, therefore, it is unlikely that a liquid phase plays an important role in the SHS. Using lattice parameters and known thermal expansion data, the ignition temperature was estimated at 901 ± 8°C and the combustion temperature at 2320 ± 50°C.
Ref.:
- Riley D.P., Kisi E.H., Phelan D. - SHS of Ti3SiC2: Ignition temperature depression by mechanical activation, J. European Ceram. Soc. 26, 1051-1058 (2006).
- Riley D.P., Kisi E.H., Hansen T.C., Hewat A.W. - Self-propagating high-temperature synthesis of Ti3SiC2: I, Ultra-high-speed neutron diffraction study of the reaction mechanism, J. Am. Ceram. Soc. 85, 2417-2424 (2002).
